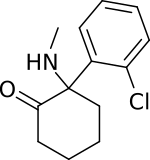
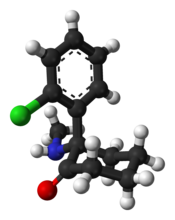
「ケタミン」の版間の差分
ナビゲーションに移動
検索に移動
細編集の要約なし |
細編集の要約なし |
||
| 24行目: | 24行目: | ||
| legal_US = Schedule III | | legal_US = Schedule III | ||
| legal_UN = Unscheduled | | legal_UN = Unscheduled | ||
| routes_of_administration = Any<ref><pubmed>28657160</pubmed></ref><ref><pubmed> 28339431 </pubmed></ref><ref name="MathewZarate2016"><pubmed>24257811 </pubmed> | | routes_of_administration = Any<ref><pubmed>28657160</pubmed></ref><ref><pubmed> 28339431 </pubmed></ref><ref name="MathewZarate2016"><pubmed>24257811 </pubmed></ref> | ||
| addiction_liability = Low–moderate<ref name="NHM-PCP and ketamine">'''Malenka RC, Nestler EJ, Hyman SE'''<br>Chapter 15: Reinforcement and Addictive Disorders |quote=Phencyclidine (PCP or angel dust) and ketamine (also known as special K) are structurally related drugs... their reinforcing properties and risks related to compulsive abuse<br>edited by Sydor A, Brown RY, Molecular Neuropharmacology: A Foundation for Clinical Neuroscience 2nd ed. pp. 374–375 (2009) McGraw-Hill Medical, New York</ref><!--Start widen drugbox--><br /> <!--End widen drugbox--> | | addiction_liability = Low–moderate<ref name="NHM-PCP and ketamine">'''Malenka RC, Nestler EJ, Hyman SE'''<br>Chapter 15: Reinforcement and Addictive Disorders |quote=Phencyclidine (PCP or angel dust) and ketamine (also known as special K) are structurally related drugs... their reinforcing properties and risks related to compulsive abuse<br>edited by Sydor A, Brown RY, Molecular Neuropharmacology: A Foundation for Clinical Neuroscience 2nd ed. pp. 374–375 (2009) McGraw-Hill Medical, New York</ref><!--Start widen drugbox--><br /> <!--End widen drugbox--> | ||
| class = [[NMDA receptor antagonists]]; [[General anesthetics]]; [[Dissociative hallucinogen]]s; [[Analgesic]]s; [[Antidepressant]]s | | class = [[NMDA receptor antagonists]]; [[General anesthetics]]; [[Dissociative hallucinogen]]s; [[Analgesic]]s; [[Antidepressant]]s | ||
2020年7月9日 (木) 22:31時点における版
- ↑
Bell, R.F., Eccleston, C., & Kalso, E.A. (2017).
Ketamine as an adjuvant to opioids for cancer pain. The Cochrane database of systematic reviews, 6, CD003351. [PubMed:28657160] [PMC] [WorldCat] [DOI] - ↑
Moyse, D.W., Kaye, A.D., Diaz, J.H., Qadri, M.Y., Lindsay, D., & Pyati, S. (2017).
Perioperative Ketamine Administration for Thoracotomy Pain. Pain physician, 20(3), 173-184. [PubMed:28339431] [WorldCat] - ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8
Frohlich, J., & Van Horn, J.D. (2014).
Reviewing the ketamine model for schizophrenia. Journal of psychopharmacology (Oxford, England), 28(4), 287-302. [PubMed:24257811] [PMC] [WorldCat] [DOI] - ↑ Jianren Mao
Opioid-Induced Hyperalgesia
CRC Press, April 2016 - ↑ 5.0 5.1 5.2 Pascal Kintz. (2014).
Toxicological Aspects of Drug-Facilitated Crimes
Elsevier Science - ↑
Molero, P., Ramos-Quiroga, J.A., Martin-Santos, R., Calvo-Sánchez, E., Gutiérrez-Rojas, L., & Meana, J.J. (2018).
Antidepressant Efficacy and Tolerability of Ketamine and Esketamine: A Critical Review. CNS drugs, 32(5), 411-420. [PubMed:29736744] [WorldCat] [DOI] - ↑ 7.0 7.1 7.2
Sinner, B., & Graf, B.M. (2008).
Ketamine. Handbook of experimental pharmacology, (182), 313-33. [PubMed:18175098] [WorldCat] [DOI] - ↑
Hashimoto, K. (2019).
Rapid-acting antidepressant ketamine, its metabolites and other candidates: A historical overview and future perspective. Psychiatry and clinical neurosciences, 73(10), 613-627. [PubMed:31215725] [PMC] [WorldCat] [DOI] - ↑
Alan F. Schatzberg, Charles B. Nemeroff. (2017)
The American Psychiatric Association Publishing Textbook of Psychopharmacology, Fifth Edition
American Psychiatric Pub, pp550-. - ↑ 10.0 10.1
Zhang, K., & Hashimoto, K. (2019).
An update on ketamine and its two enantiomers as rapid-acting antidepressants. Expert review of neurotherapeutics, 19(1), 83-92. [PubMed:30513009] [WorldCat] [DOI] - ↑ Andrew Dickman, Jennifer Schneider. (2016)
The Syringe Driver: Continuous Subcutaneous Infusions in Palliative Care
Oxford University Press, pp. 114- - ↑ Frank J. Dowd, Bart Johnson, Angelo Mariotti. (2016)
Pharmacology and Therapeutics for Dentistry – E-Book
Elsevier Health Sciences, pp235– - ↑
Hijazi, Y., & Boulieu, R. (2002).
Contribution of CYP3A4, CYP2B6, and CYP2C9 isoforms to N-demethylation of ketamine in human liver microsomes. Drug metabolism and disposition: the biological fate of chemicals, 30(7), 853-8. [PubMed:12065445] [WorldCat] [DOI] - ↑
Quibell, R., Prommer, E.E., Mihalyo, M., Twycross, R., & Wilcock, A. (2011).
Ketamine*. Journal of pain and symptom management, 41(3), 640-9. [PubMed:21419322] [WorldCat] [DOI] - ↑ I. K. Morton, Judith M. Hall. (2012)
Concise Dictionary of Pharmacological Agents: Properties and Synonyms
Springer Science & Business Media, pp159–